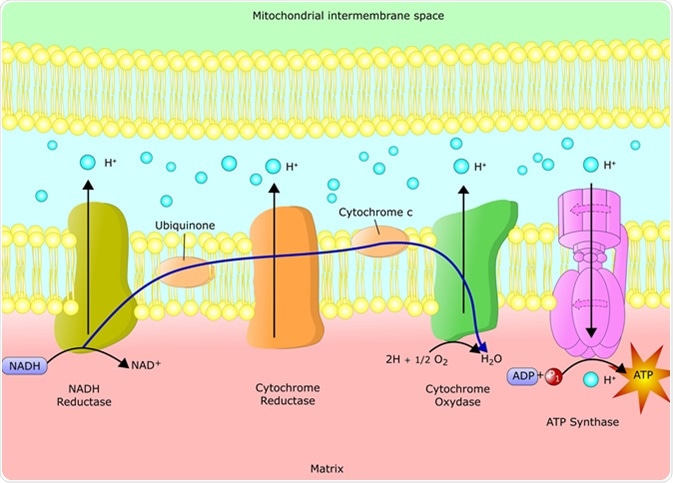
The final electron acceptor in the electron transport chain is the oxygen.Electrons in the electron transport chain are transferred from molecules of lower redox potential to that of higher redox potential.The electron transport chain consists of 5 complexes present in a sequence that carry out the transfer of electrons and coupled ATP production.They are also called ‘powerhouses of the cell’. 
In eukaryotes, mitochondria is the site for aerobic respiration.This backflow of protons results in thermogenesis instead of ATP production. These uncouplers allow for the flowing back of protons into the mitochondrial matrix. Thyroxine is also a natural uncoupler of the process. This can be done with the help of uncoupling protein - thermogenin. But sometimes, uncoupling these two processes can serve other biological purposes. In this step, eight protons are transferred from the mitochondrial matrix.Įlectron Transport Chain and Oxidative Phosphorylationįor the production of ATP, it is necessary to couple the electron transport chain with oxidative phosphorylation. This reaction results in the formation of two water molecules. In this, four electrons from cytochrome c are transferred to oxygen molecules (O2). It is also called cytochrome AA3 complex. During this transfer, four protons are transferred across the membrane. Cytochrome c is found in the intermembrane space. Two electrons from QH2 are transferred to cytochrome c. This is also called CoQH2 - cytochrome c reductase. In this step, transfer of protons does not take place.Įlectron Transfer Chain Complexes Complex III - Cytochrome bc1 Complex In this step, electrons are transferred from succinate to coenzyme Q (or Ubiquinone) via FAD (flavin adenine dinucleotide). This complex is also called succinate-CoQ reductase. During this, four protons (H+) are transferred across the membrane. As the first step of the electron transport chain, two electrons from NADH are transferred to ubiquinone (Q) via a Fe-S molecule, reducing it to ubiquinol (QH2). The first complex in the electron transport chain is the NADH Ubiquinone Oxidoreductase. The complexes involved in the electron transport and its process is given below: Complex I - NADH Ubiquinone Oxidoreductase Oxygen is the final electron acceptor in aerobic respiration. This transfer of electrons from lower to higher redox potential continues until it reaches the final electron acceptor. In the electron transport chain, electrons from NADH and FADH2 are transferred to an electron acceptor having a higher redox potential. Since the production of ATP occurs in the mitochondria, they are called ‘powerhouses of the cell’.ĭifference between Prokaryotic and Eukaryotic Transcription Mitochondria contains their own DNA and ribosomes, so they are called semi-autonomous. Matrix - Mitochondrial matrix is the space enclosed by the membranes of mitochondria. Cristae serves to increase the surface area of the membrane. Intermembrane Space - The space present between the outer and the inner membrane is called the intermembrane space.Ĭristae - The inner membrane of the mitochondria folds inside to give rise to structures called cristae. It is impermeable to most of the molecules. Inner membrane - It is the membrane which contains many protein complexes involved in ETS and ATP production. The outer membrane also contains enzymes. Outer membrane - This is the outermost membrane of mitochondria and consists of channels called porins that allow some molecules to cross the membrane. They are small in size and consists of the following: Mitochondria are the sites of aerobic respiration in eukaryotic cells. Mitochondria are the double-membrane bound cell organelles found in most eukaryotic cells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
